Transplantation Direct
JUST RELEASED - TRANSPLANTATION DIRECT - AUGUST ISSUE
The August issue content of Transplantation Direct is ready for viewing. A variety of topics are covered, including how prior donor sex can affect females undergoing re-transplantation. Other donor issues are studied that examine factors influencing transplant recipients decisions to accept organs from HCV viremic donors, and pilot testing of an automated donor organ referral system to increase the donor organ pool. This month, we especially feature articles in liver transplantation, including an algorithmic intra-operative assessment of arterial inflow involving median arcuate ligament restrictions, and a comparison of conservative versus endovascular intervention to reduce posttransplant ascites. Also, a novel telemedicine method is tested to remotely obtain frailty data via the 6-minute walk test in liver transplant candidates, and the value of accurate reporting of concurrent liver disease is supported by data being collected in Australia and New Zealand. Please take some time to visit the Transplantation Direct website for full details and open access to these articles.
Transplantation Updates

Transplantation - Online First Article
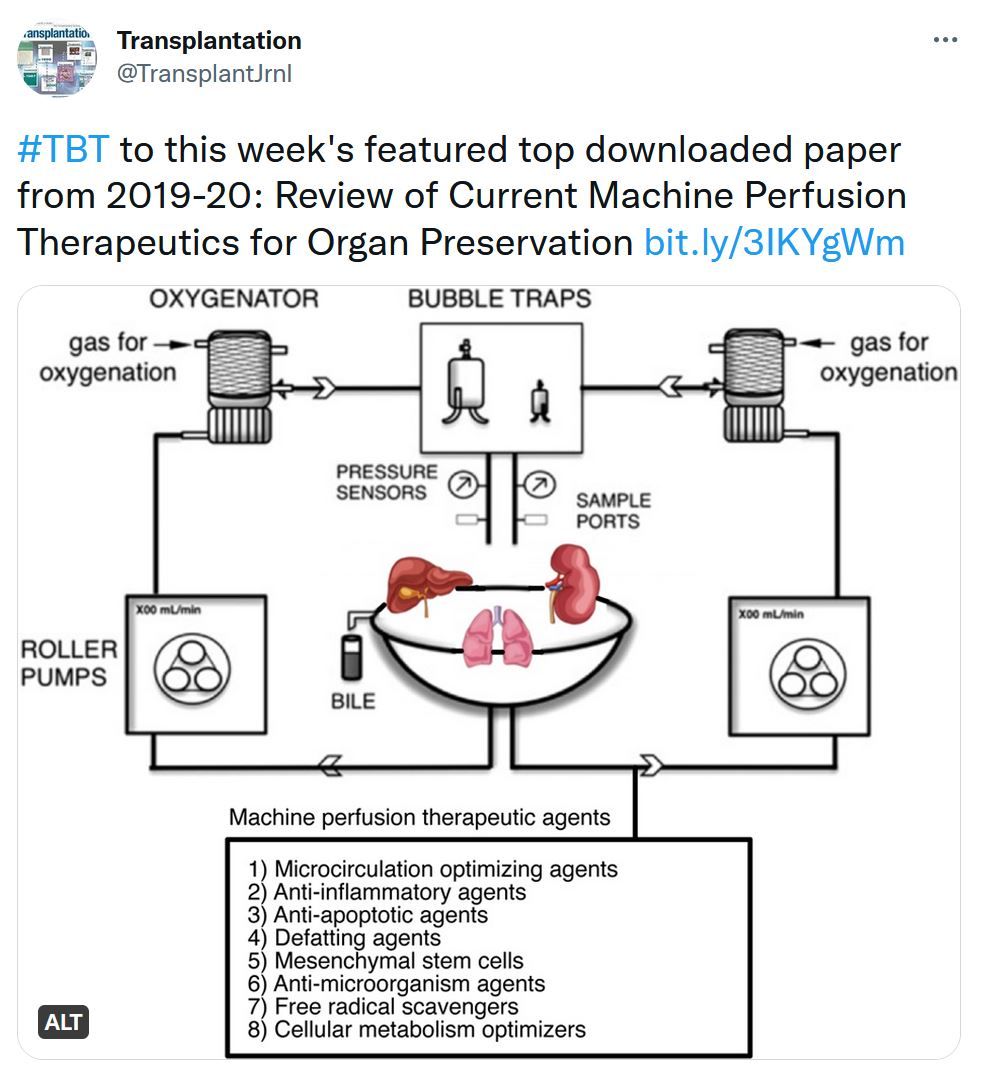
Review of Current Machine Perfusion Therapeutics for Organ Preservation
Because of the high demand of organs, the usage of marginal grafts has increased. These marginal organs have a higher risk of developing ischemia-reperfusion injury, which can lead to posttransplant complications. Ex situ machine perfusion (MP), compared with the traditional static cold storage, may better protect these organs from ischemia-reperfusion injury. In addition, MP can also act as a platform for dynamic administration of pharmacological agents or gene therapy to further improve transplant outcomes. Numerous therapeutic agents have been studied under both hypothermic (1–8°C) and normothermic settings. Here, we review all the therapeutics used during MP in different organ systems (lung, liver, kidney, heart). The major categories of therapeutic agents include vasodilators, mesenchymal stem cells, antiinflammatory agents, antiinfection agents, siRNA, and defatting agents. Numerous animal and clinical studies have examined MP therapeutic agents, some of which have even led to the successful reconditioning of discarded grafts. More clinical studies, especially randomized controlled trials, will need to be conducted in the future to solidify these promising results and to define the role of MP therapeutic agents in solid organ transplantation.Transplantation Direct - Highlighted Tweet
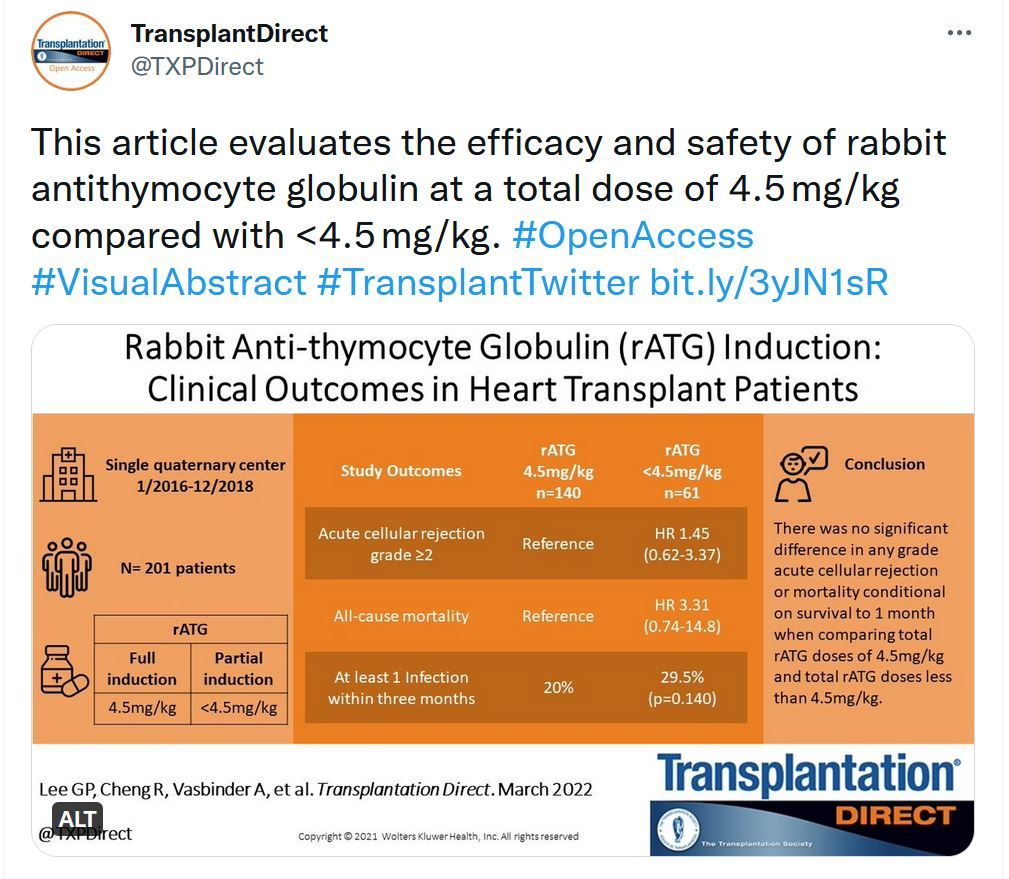
Retrospective Evaluation of Rabbit Antithymocyte Globulin Induction in Heart Transplant Patients
The dosing intensity of antithymocyte globulin as induction therapy in heart transplantation remains controversial. We sought to evaluate the efficacy and safety of rabbit antithymocyte globulin at a total dose of 4.5 mg/kg compared with <4.5 mg/kg.Women in Transplantation & Asian Society of Transplantation - Fellowship Grant

TTS Needs Assessment Survey
The slogan we adopted for the TTS 2022 Congress is "Committed to Access and Transparency" and as a lead-up to the Congress we have launched a new needs assessment survey which will aid TTS in developing new programs, educational material and strategies to better improve access and transparency in the field of transplantation. As a non-governmental organization in official relations with the World Health Organization (WHO) the information collected will feed into the work TTS is doing as part of our WHO collaboration.
Your support, by completing this survey, will contribute to the success of this initiative. Your answers will be kept confidential and anonymous.
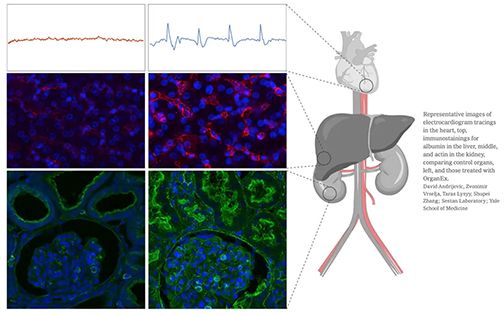
Cell reanimation in the organs of dead pigs confounds conventional understanding of life and death
Contact
Address
The Transplantation Society
International Headquarters
740 Notre-Dame Ouest
Suite 1245
Montréal, QC, H3C 3X6
Canada
Используйте Вавада казино для игры с бонусом — активируйте промокод и начните выигрывать уже сегодня!