Members were emailed their invoices two weeks ago.
If you are not a member click the Join TTS button below!
Latest Video Content Posted

Videos from the Congress are now available to TTS Members.

SPLIT 2020 Meeting videos are now available to TTS & SPLIT Members.

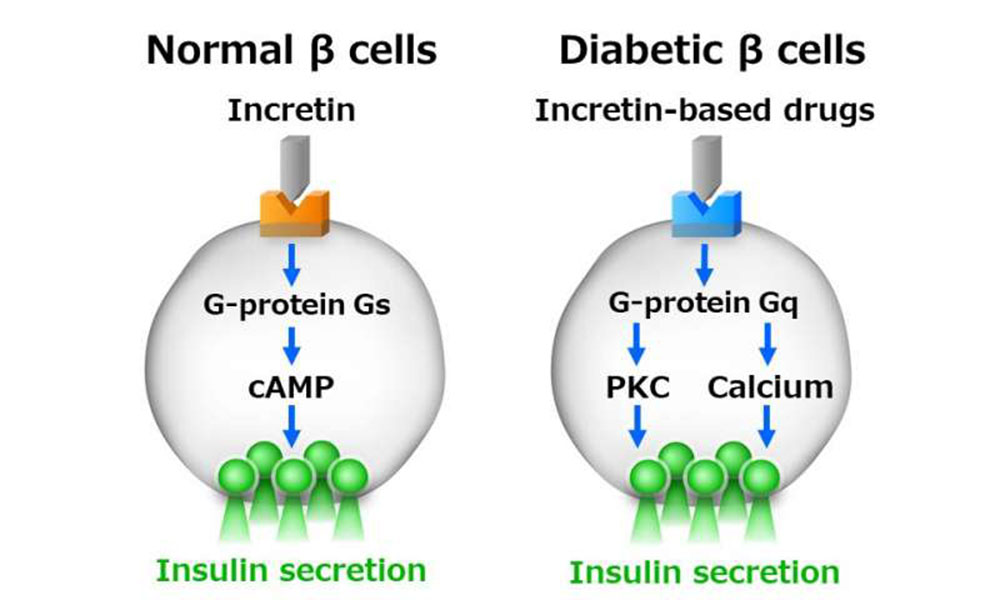
Beta Cells Summit videos are now available to TTS & IPITA Members.
Nov 22 - FUNDAMENTALS TO COMPLEX DECISIONS WITH KIDNEY TRANSPLANTATION - Milagros Samaniego Picota
Click to view (TTS Members only)Nov 19 - Tips and Tricks for Virtual Care in Pediatric Transplant - IPTA Allied Health and Nursing Professionals Webinar - Ashley Graham, Julie Guillen, Lisa Remaley, Robin Deliva, Anna Galloway, Rosa Reed-Berendt
Click to view (IPTA or TTS Members only) TTS-ILTS Paired Transplant Centers Program
Application Deadline - January 1, 2021
The TTS-ILTS Paired Transplant Centers Program is a collaboration between The Transplantation Society (TTS) and the International Liver Transplantation Society (ILTS) supporting new liver transplant programs in emerging economies.
TTS and TTS Sections News

«HOT OFF THE PRESS»
RECENT PUBLICATIONS IDENTIFIED BY TTS EDUCATION COMMITTEE ON COVID-19
Selected Publications by TTS Education Committee. This week's selection made by Enver Akalin, Gabriel Gondolesi and Millie Samaniego
CORONAVIRUS (COVID-19) UPDATES
IN THE NEWS
UPCOMING MEETINGS AND ANNOUNCEMENTS
Contact
Address
The Transplantation Society
International Headquarters
740 Notre-Dame Ouest
Suite 1245
Montréal, QC, H3C 3X6
Canada
Используйте Вавада казино для игры с бонусом — активируйте промокод и начните выигрывать уже сегодня!